Molar mass of carbon dioxide11/27/2023  The molar mass of benzene is therefore 78g/mol The elements in C 6H 6 and their relative atomic masses are: C = 12, H = 1 Therefore, the molar mass of salicylic acid is 138g/mol The elements in C 7H 6O 3 and their relative atomic masses are: C = 12, H = 1, O = 16Ĭalculating its molar mass: 12x7 + 1圆 + 16x3 Therefore, the molar mass of acetone is 58g/mol The elements in C 3H 6O and their relative atomic masses are: C = 12, H = 1, O = 16Ĭalculating its molar mass: 12x3 + 1圆 + 16 The relative atomic masses of elements in NaCl: Na = 23, Cl = 35.5Ĭalculating its molar mass, we have 23 + 35.5 = 58.5 Therefore, the molar mass of carbon(IV) oxide is 44g/mol The relative atomic masses of the elements in CO 2: C = 12, O = 16Ĭalculating its molar mass, we have 12 + 16x2 Carbon(IV) oxide, also called carbon dioxide, CO 2 The molar mass of ethanol is therefore 46g/molġ7. The relative atomic masses of elements in C 2H 5OH are: C = 12, H = 1, O = 16Ĭalculating its molar mass = 12x2 + 1圆 + 16 Relative atomic masses of elements in CH 3OH are: C = 12, H = 1, O = 16Ĭalculating the molar mass, we have, 12 + 1x4 + 16 Therefore, the molar mass of CaCl 2 is 111g/mol Relative atomic masses of elements in CaCl 2 are: Ca = 40, Cl = 35.5Ĭalculating its molar mass, we have, 40 + 35.5x2 The molar mass of KCl is therefore 74.5g/mol The relative atomic masses of elements in KCl are: K = 39, Cl = 35.5Ĭalculating its molar mass, 39 + 35.5 = 74.5 Therefore, the molar mass of CuSO 4 is 160g/mol The relative atomic masses of the elements in CuSO 4 are: Cu = 64, S = 32, O = 16Ĭalculating the molar mass, we have: 64 + 32 + 16x4 Therefore, the molar mass of butane is 58g/mol The relative atomic masses of elements in C 4H 10 are: C = 12, H = 1 Therefore, the molar mass of NH 3 is 17g/mol The relative atomic masses of the elements in NH 3 are: N = 14, H = 1 Therefore, the molar mass of sodium hydroxide is 40g/mol The relative atomic masses of the elements in NaOH are: Na = 23, O = 16, H = 1Ĭalculating the molar mass of NaOH: 23 + 16 + 1 = 40 Therefore, the molar mass of acetic acid is 60g/mol 
The relative atomic masses of elements in CH 3COOH: C = 12, H = 1, O = 16Ĭalculating the molar mass of CH 3COOH, we have 12x2 + 1x4 + 16x2 The relative atomic masses of the elements in CH 4 are: C = 12, H = 1Ĭalculating the molar mass gives 12 + 1x4 Therefore, the molar mass of sucrose is 342g/mol The relative atomic masses of elements in C 12H 22O 11 are: C = 12, H = 1, O = 16Ĭalculating the molar mass, we have 12x12 + 1x22 + 16x11 Therefore, the molar mass of CaCO 3 is 100g/mol The relative atomic masses of elements in CaCO 3 are Ca = 40, C = 12, O = 16 Therefore, the molar mass of glucose is 180g/mol The relative atomic masses of elements in C 6H 12O 6Ĭalculating the molar mass, we have: 12圆 + 1x12 + 16圆 The relative atomic masses of the elements in H 2SO 4 are: H = 1, S = 32, O = 16.Ĭalculating the molar mass, we have: 1x2 + 32 + 16x4 The molar mass is calculated as follows: relative atomic mass of H + relative atomic mass of chlorine, which is 1 + 35.5 = 36.5g/mol The relative atomic masses of the elements in hydrochloric acid, HCl are H = 1, Cl = 35.5. The molar mass of water, H 2O is therefore 18g/mol  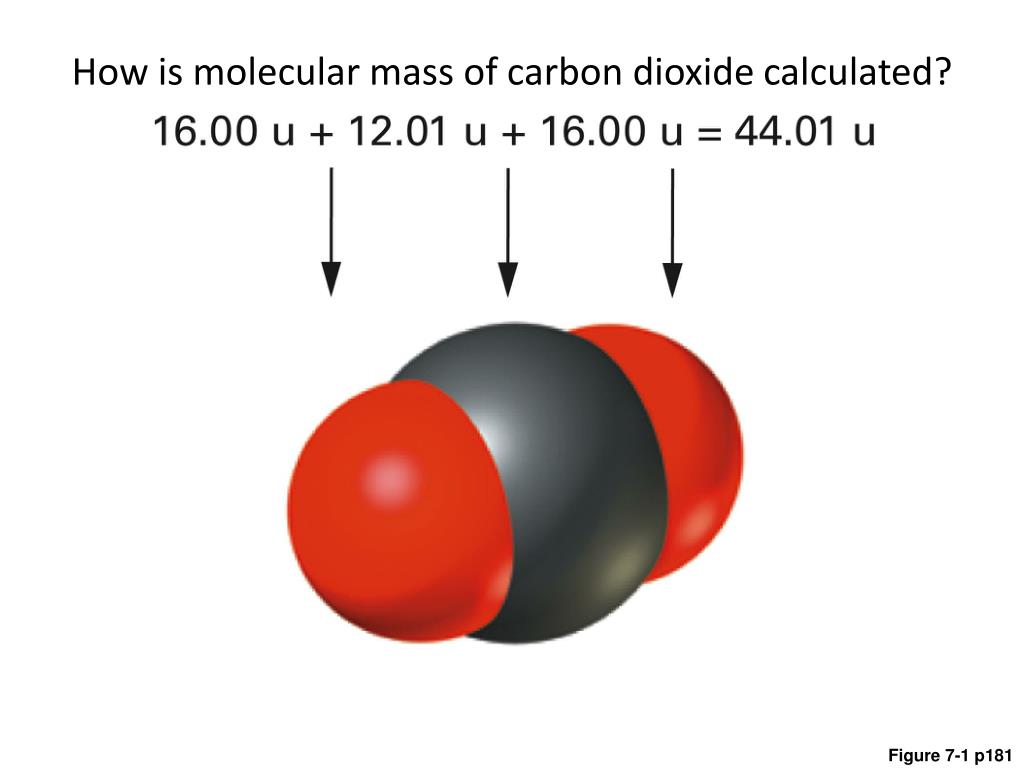 
Relative atomic mass of hydrogen x number of atoms of hydrogen + relative atomic mass of oxygen x number of atoms of hydrogen. The relative atomic masses of the elements in water, H 2O are: H = 1, O = 16. For an element, the molar mass is the atomic mass of the element.įor example: calculate the molar mass of the following substances: The molar mass of a substance is calculated by summing up the product of the relative atomic mass and number of atoms of all the elements that make up the substance as expressed in its chemical formula. It is a physical quantity that is measured and expressed in g/mol. The molar mass of a substance (chemical element or compound) is the mass of one mole of the substance. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
